O-ring 0.313 ID 0.438 OD BCG 0.062 W EPR 80 Durometer
| 0210-0691-300 | |
| New | |
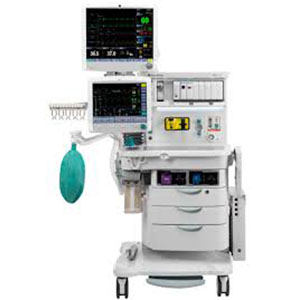
| Anesthesia Delivery | |
| GE HealthCare | |
| Datex-Ohmeda | |
| GE HealthCare | Outright |
Enter your approval number and submit to add item(s) to cart.
Please enter approval number
OR
Don't know your approval number? Call 800-437-1171
Enter opt 1 for the first three prompts, and have your System ID available.
If you add item(s) to cart and submit your order without the
approval number, GE will contact you before your order
can be confirmed for shipment.
Select your approver's name and submit to add item(s) to your cart
Please Select Approver Name
OR
Don't know your approval number? Call 800-437-1171
Enter opt 1 for the first three prompts, and have your System ID available.
If you add item(s) to cart and submit your order without
selecting an approver, GE will contact you before your order
can be confirmed for shipment.
Features
- High temperature resistance
- Resistant to acids, oxygen, fuels, mineral oils, aromatics and hydraulic fluids and many organic solvents and chemicals over a wide temperature range
- Excellent resistance to aging and ozone
- Low gas permeability